Ohh-med Medical Ltd. is committed to clinical research and to collaboration with medical professionals and researchers worldwide, aiming to demonstrate scientifically the efficacy of our products.

A pilot clinical study was performed to assess the efficacy of the Vertica®, our RF home–use device to improve penile erections.
The study, done at the Neuro-urology unit, Rambam Healthcare Campus, was conducted on 28 patients with erectile dysfunction and showed significant improvement and enhancement of penile erections.
It also demonstrated a high sexual quality of life and high satisfaction from treatment.

In this study, the patients that were on Erectile Dysfunction (ED) treatment were recruited as well and were requested to remain under their current medical treatment. This way we were able to assess the net effect of RF treatment in naïve as well as in treated patients. We included men with organic ED for a least 6 months and that had a stable relationship.
We excluded patients with premature ejaculation and with Peyronie’s disease, and patients with pelvic malignancy within the past 3 years. We determined the severity of ED using the validated erectile function domain of the International Index of Erectile function questionnaire (IIEF-EF) and included only those with a score of 11-20 points, which means that only moderate and moderate to mild ED patients were included.
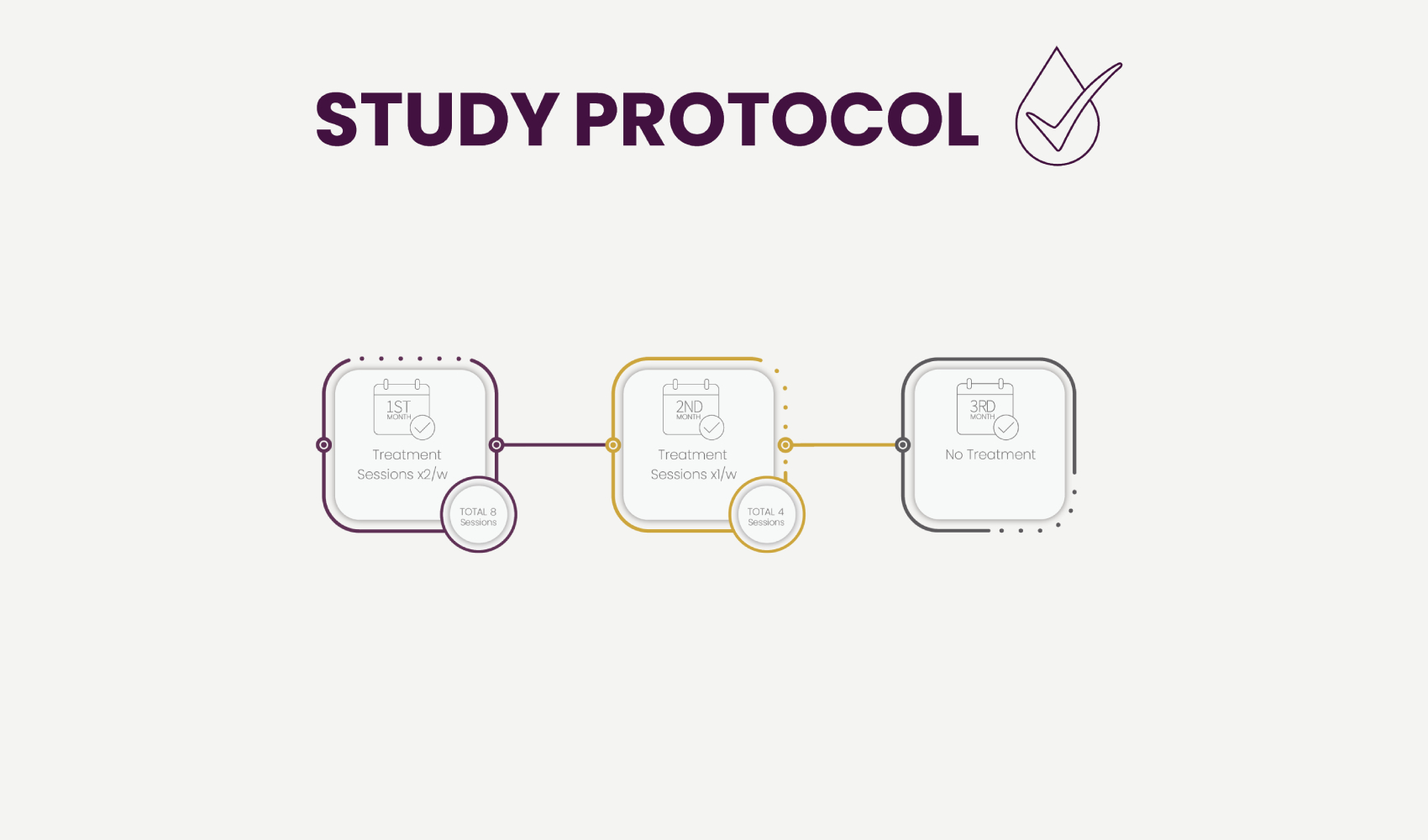
Each patient received 12 treatments, twice a week during 4 weeks and once a week for another 4 weeks. Treatments were self- applied by the patients themselves after receiving a short instruction session.
We evaluated the patients’ outcome by the same IIEF-EF questionnaire before treatment and one month after end of treatment.
To assess their quality of sexual life and their satisfaction from treatment, we handed out validated questionnaires one month after completing the treatment.
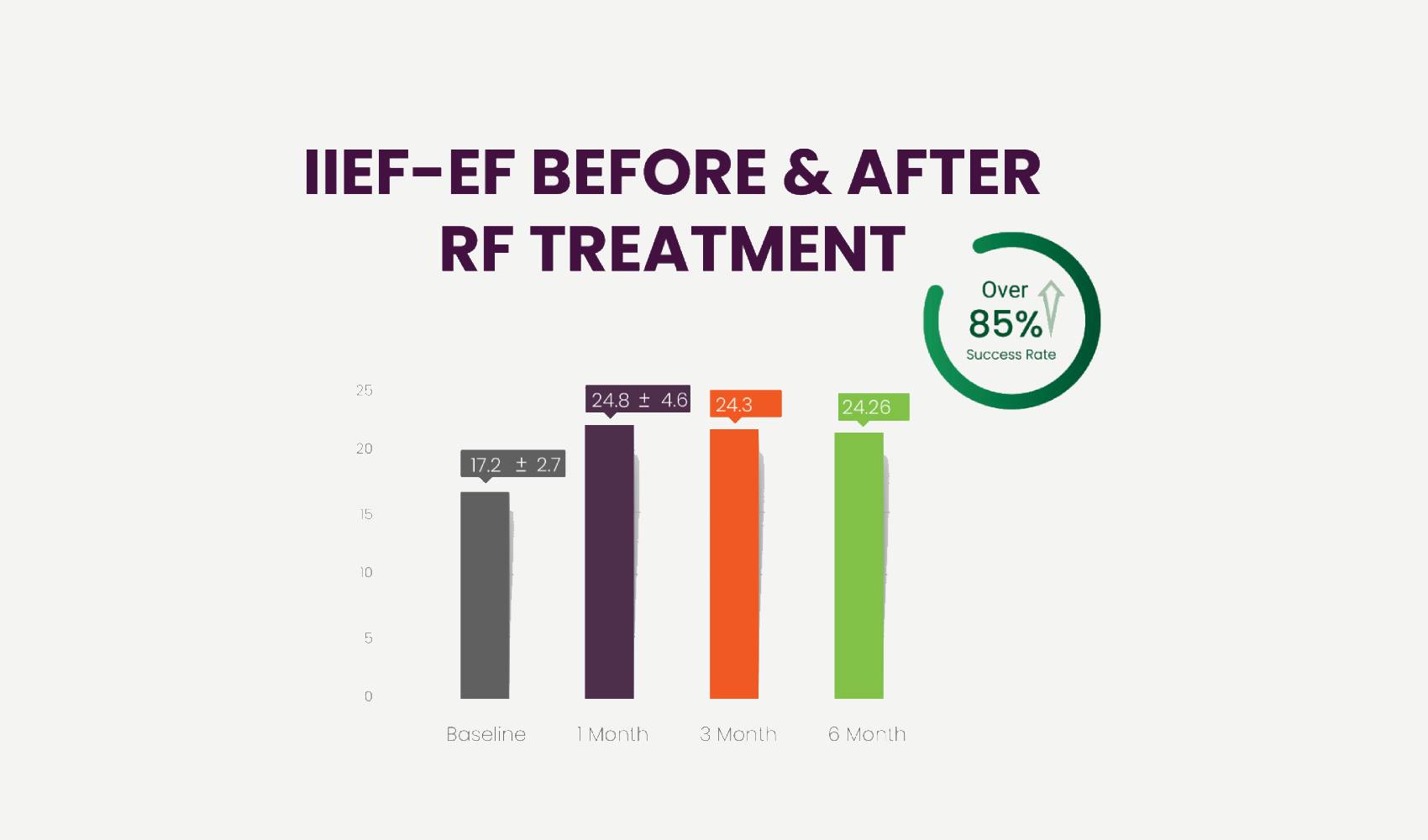
Our results showed that 25/28 patients (89%) achieved an improvement in their erectile function, expressed by an increase of minimum 5 points for the moderate ED patients and a minimum increase of 2 points for the mild ED cases. Overall, an average increase of 7.6 points in the IIEF-EF questionnaire was achieved, which is an impressive increase representing a significant enhancement of erectile function.

The Edits questionnaire (Erectile Dysfunction Index of treatment Satisfaction) reached an average final score of 76.8 ± 20.3 (>50=satisfied) and only 4/28 (14.2%) were below 50 points.
The SQOL questionnaire (Sexual Quality of Life) reached an average final score of 67.6 ± 29.4 (>60 =positive SQOL), 10/28 (36%) were below 60.
The study is limited to 28 patients and is not a placebo controlled study, but we are now in the process of a follow up of one year to examine the duration of the effect.
On our first 10 patients we performed and objective assessment of the erectile mechanism (penile FMD- Flow Mediated Dilatation) and demonstrated an improvement of all 3 parameters of this test in 9/10 patients, indicating a real improvement in blood flow of the penis, strengthening our hypothesis that RF instigates a physiologic effect on the erectile mechanism.
Our results showed that 25/28 patients (89%) achieved an improvement in their erectile function, expressed by an increase of minimum 5 points for the moderate ED patients and a minimum increase of 2 points for the mild ED cases.
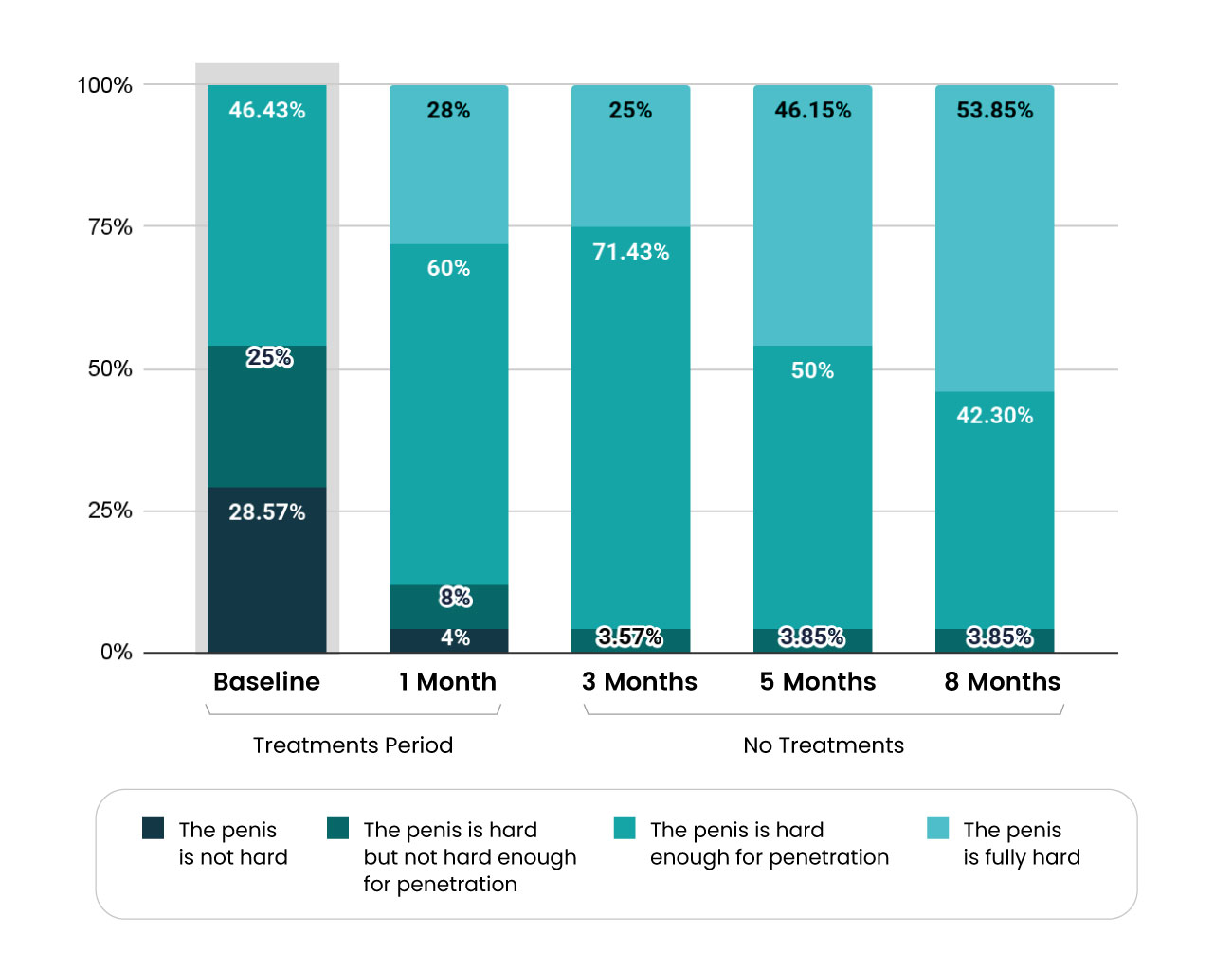
One of the exciting results coming from the first clinical research performed on the treatment of erectile dysfunction using radio waves (RF) is the indication of an improvement in erection hardness score during treatment with the Vertica device and maintenance of a high level of hardness for at least six months from the time of the last treatment with the device.
One of the accepted ways of assessing the hardness of the erection is via an easy to use measurement tool known as the EHS (Erection Hardness Score). The EHS measurement ranks the hardness of the erection on a scale of one to four, where four is the maximum score. The language used is simple and direct, such that men with erectile dysfunction can use the scale in order to assess the severity of their condition and monitor the effect of the treatment.
For full information about the results of the EHS measurement »
No side effects were reported by any of our users, other than a gentle warm sensation and slight redness. Both of these are temporary and should go away after the treatment with Vertica® device. You can set the level of RF energy emitted by the device for maximum comfort.
As any medical process, different bodies respond differently to treatments.
Usually, men start to notice an improvement within the first two weeks of treatment; some even experience ‘morning wood’ and spontaneous erections throughout the day. Other men notice improvement after 4-8 weeks of treatment.
Vertica® is the first non-invasive, drug-free device in the world to effectively treat erectile issues and enhance erections. It is entirely safe for use and was proven to be effective in clinical trials. Treatments with Vertica® don’t require a prescription or a doctor’s visit. They are fully discreet, pain-free, and have no known side effects. You can do them by yourself, at home and anytime.
Enter your information and we'll be in touch soon.